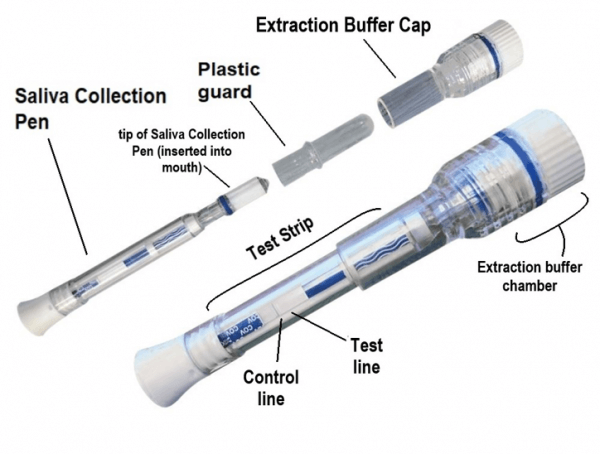
COVID-19 Antigen Rapid Saliva Test Pen
The Rapid Response® COVID-19 Antigen Rapid Test Pen (Saliva) is an in vitro immunoassay. It is the World’s First ‘No-Spit’ Saliva Antigen Rapid Test Pen for SARS-CoV-2! The COVID-19 antigen rapid test pen is for the direct and qualitative detection of SARS-CoV-2 viral nucleoprotein antigens from saliva samples through a visual interpretation of colour development. They are manufactured in China and are extremely user-friendly for rapid results in 15 minutes. These antigen rapid test pens are available at a low-cost and can be shipped immediately.
Key Benefits:
-
- First no-spit test that is COVID-19 dedicated – has a sponge-like tip on the pen that can collect a sufficient volume of specimen in 2 minutes without the discomfort of a nasal/throat swab
- All in One – There is no time wasted setting up a workstation and no need to handle numerous assay components as all that is needed is one pen that can be used at any location
- Manufactured in China – The product is manufactured in China and comes at a low-cost with availability for immediate shipment
- Ease of Use – The rapid antigen test pens are extremely easy to use and they require little set up as they are an all in one product
- Rapid Results – The user can expect results in just 15 minutes of taking the antigen test
How It Works:

* These antigen rapid test pens are for professional use only and should be administered by a trained healthcare professional
Bring devices to room temperature (18-30°C) before use.
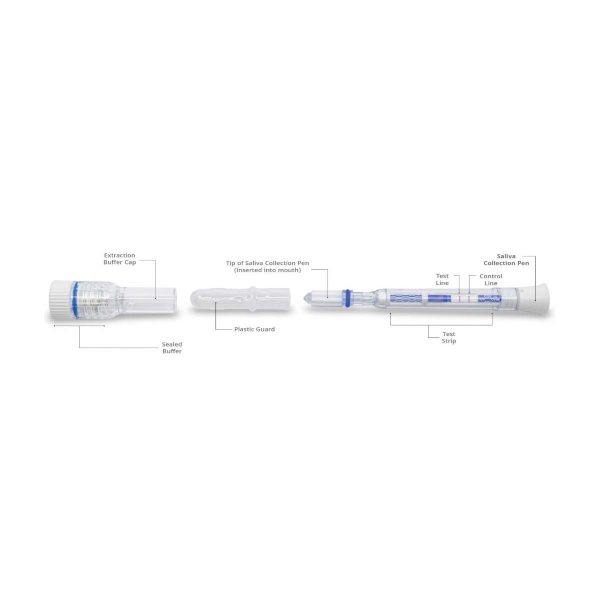
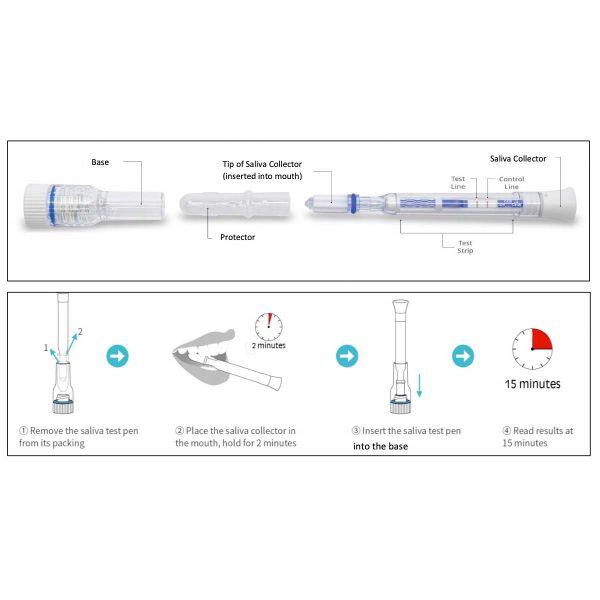
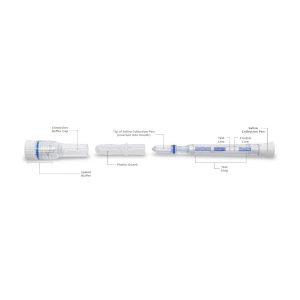
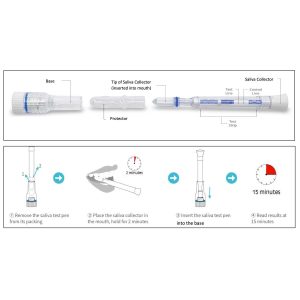
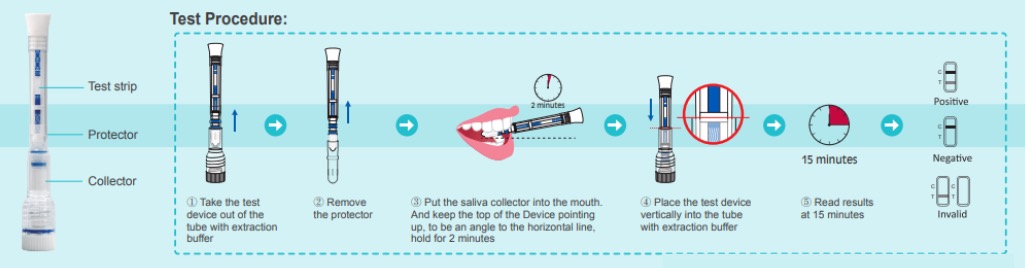
- Open the pouch and remove the Saliva Collection Pen and Extraction Buffer Cap from the packaging. Label the Saliva Collection Pen with patient identification. For best results, the assay should be performed within two hours
- Clear throat, then cough deeply four times with mouth closed before collecting the samples
- Remove the plastic guard from the tip of the Saliva Collection Pen
- Place the Saliva Collection Pen in the mouth above the tongue. Hold in place for 2 minutes
- Remove the Saliva Collection Pen from the patient’s mouth. Holding the Saliva Collection Pen vertically, place it into the Extraction Buffer Cap. Press down to break the seal of the Extraction Buffer Cap, which will release the buffer solution
- Start the timer immediately after breaking the seal. Read the results at 15 minutes
Test Principle:
Anti-SARS-CoV-2 antibodies are immobilized on the test region of the nitrocellulose membrane. These antibodies are conjugated to coloured particles that are immobilized on the conjugated pad. A sample is added to the extraction buffer which is optimized to release the SARS-CoV-2 antigens from specimen. During testing, target antigens, if present in the saliva samples, will be released into the extraction buffer individually packed in the kit. Consequently, the extracted antigens will bind to anti-SARS-CoV-2 antibodies conjugated to coloured particles.
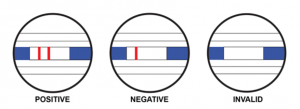
As the specimen migrates along the strip by capillary action and interacts with reagents on the membrane, the complex will be captured by the anti-SARS-CoV-2 antibodies at the test region. Excess coloured particles are captured at the internal control zone. The presence of a coloured band in the test region indicates a positive result for the SARS-CoV-2 viral antigens, while its absence indicates a negative result. A coloured band at the control region serves as a procedural control, indicating that the proper volume of specimen has been added and membrane wicking is working.
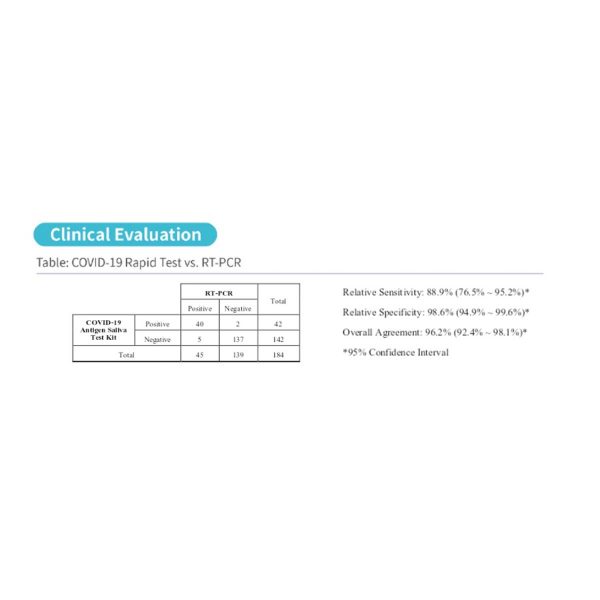
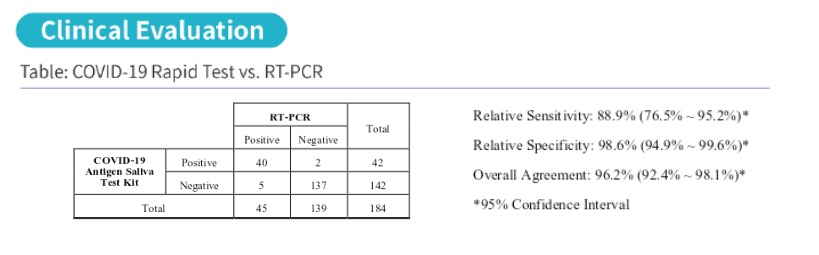
Clinical Performance Data:
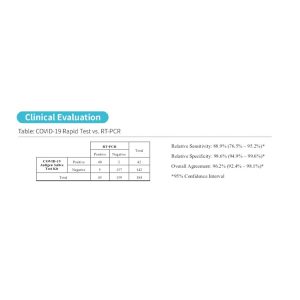
These COVID-19 Antigen Rapid Test Pens have been clinically tested in Switzerland. The results are shown below:
| COVID-19 Antigen Rapid Test Confidence Interval: | |
| Relative Sensitivity | 90.0% (69.9% – 97.2%) * |
| Relative Specificity | 97.0% (89.6% – 99.2%) * |
| Overall Agreement | 95.3% (88.6% – 98.2%) * |
| *95% Confidence Interval |
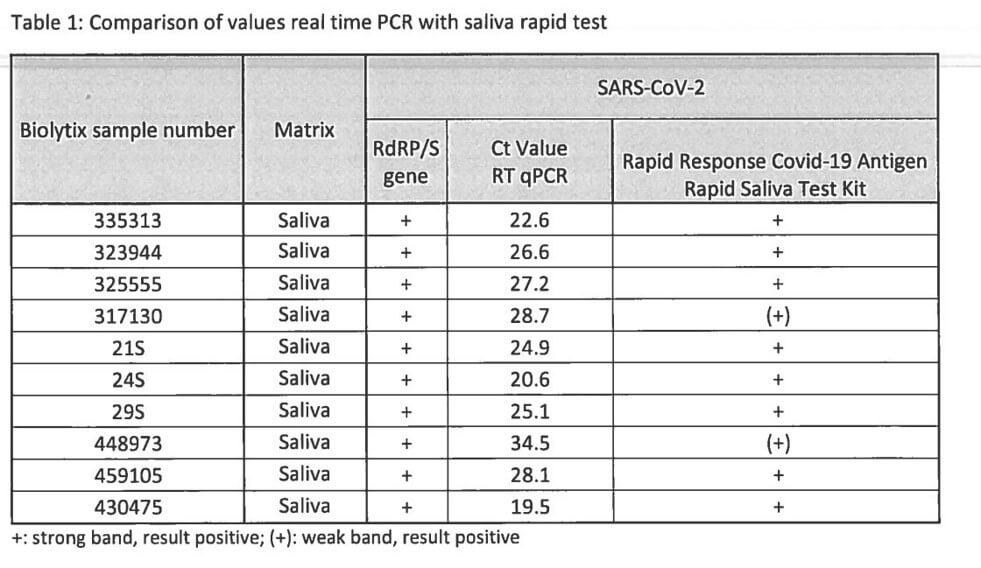
These Antigen Rapid Test Pens were also compared to real time PCR tests. According to ‘Regulation 3 on Measures to Control Coronavirus (Covid-19) (Covid-19 Regulation 3) of 19 June 2020 (as of 15 March 2021’, Chapter 3: ‘Criteria for clinical validation of Sars-CoV-2 rapid tests for professional use according to the screening standard and of Sars-CoV-2 self-tests-, the sensitivity needs to be at least 80% for the antigen rapid test pens to be available for use. The results can be seen below which demonstrates that the sensitivity of these pens is 100% accurate when compared with the real time PCR values:
Available Downloads:
- View the TGA Approval and other information PDF (114KB)
- COVID-19 Antigen Rapid Test Pen Brochure PDF (1.6MB)
- Annex 6 – Antigen Saliva Test Kit Instruction For Use PDF (357KB)
- Antigen Saliva Pen Fact Sheet PDF (1.7MB)
- Form 1 – COVID-19 Testing Testing Record Form PDF (246 KB)
- Form 2 – COVID-19 Testing Training Record Form PDF (246 KB)
Materials Provided:
Individually packaged saliva collection pen and extraction buffer cap in the same foil pouch, can come in a 5 pack or 20 pack depending on your needs.